Chemical equation balancer online
If no coefficients are required to balance the equation, simply place the integer 1 in front of each reactant and/or product.Ĭlick "Check" to see whether your response below is the correct balanced chemical equation.ģ. Type integer coefficients in front of the reactants and products to balance the chemical equations. In the following examples, you will be given the chemical equations and states. Identify the states of matter: use the following subscripts for the compounds: (g) for gaseous substances, (s) for solids, (l) for liquids, and (aq) for aqueous solutions. Finally, write out the products on the right hand side of the equation using chemical nomenclature.īalance the equation: use the Law of Conservation of Mass to balance, or achieve the same number of atoms of every element on each side of the equation.

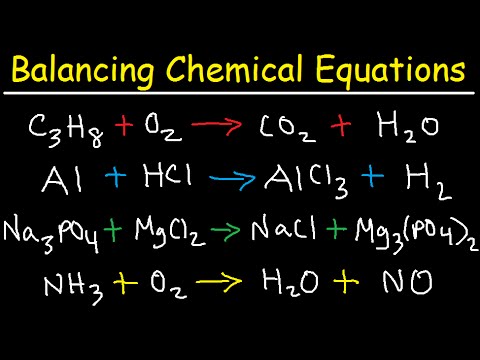
A chemical equation is a symbolic representation of a chemical reaction. The direction of the arrow indicates the flow of the reaction. Chemical Equations Putting chemical changes into words 2. Draw an arrow to represent "forms to react". Remember to use uppercase and lowercase letters, where appropriate. Enter Chemical Equation: Examples of Equations you can enter: KMnO4 + HCl KCl + MnCl2 + H2O + Cl2. Write the unbalanced equation: using chemical nomenclature (see the Periodic Table of Elements ), write out the reactants on the left hand side of the equation. Use this Calculator to balance Chemistry Equations. To balance chemical equations, the following three steps are used: A chemical reaction is a process that leads to the chemical transformation of one set of chemical substances to another. Balancing a chemical equation is a mathematical approach used in chemistry to establish the relationship between the quantity of reactants and products. The equation shows the reactants on the left hand side and the products on the right hand side of the arrow. A chemical equation describes the changes that occur during a chemical reaction.
